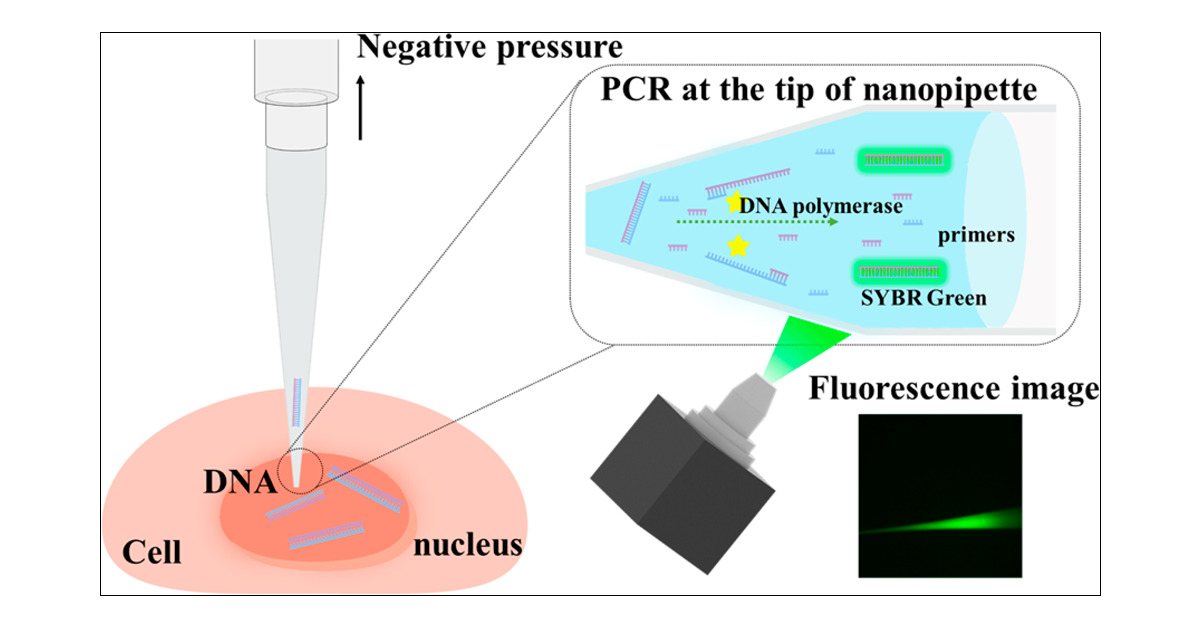
TriplePath Fluorodiagnosis
Discover comprehensive diagnostics with TriplePath Fluorodiagnosis, offering accurate and rapid testing for Ureaplasma Urealyticum, Neisseria gonorrhoeae, and Chlamydia Trachomatis through advanced Fluorescent Polymerase Chain Reaction technology.
Ureaplasma Urealyticum Fluorescent Polymerase Chain Reaction Diagnostic Kit
Unlock precise detection capabilities with our Fluorescent Polymerase Chain Reaction (PCR) Diagnostic Kit for Ureaplasma Urealyticum. Leveraging cutting-edge technology, this kit ensures swift and accurate identification of Ureaplasma Urealyticum DNA, providing crucial insights for effective diagnosis and treatment.
Neisseria gonorrhoeae Fluorescent Polymerase Chain Reaction Diagnostic Kit
Experience advanced diagnostics with our Neisseria gonorrhoeae Fluorescent Polymerase Chain Reaction (PCR) Diagnostic Kit. Designed for unparalleled sensitivity, this kit enables rapid and reliable detection of Neisseria gonorrhoeae DNA, empowering healthcare professionals with precise information for prompt intervention and patient care.
Chlamydia Trachomatis Fluorescent Polymerase Chain Reaction Diagnostic Kit
Revolutionize diagnostics with our Chlamydia Trachomatis Fluorescent Polymerase Chain Reaction (PCR) Diagnostic Kit. Engineered for high-performance accuracy, this kit facilitates the swift and specific identification of Chlamydia Trachomatis DNA, supporting clinicians in making informed decisions for optimal patient management.
Revolutionize Detection with the Ureaplasma Urealyticum Fluorescent PCR Diagnostic Kit:
Embark on a transformative journey in diagnostics with our cutting-edge Ureaplasma Urealyticum Fluorescent Polymerase Chain Reaction (PCR) Diagnostic Kit. This kit, adorned with both CE and NMPA certifications, signifies a leap forward in precision medicine, particularly addressing the challenges posed by Ureaplasma urealyticum infections.
Distinctive Features:
- Certification Distinction: Elevate confidence in diagnostic outcomes with a kit that not only meets but exceeds stringent quality standards, holding both CE and NMPA certifications.
- Targeted Detection: Unravel the mysteries of Ureaplasma urealyticum infections with a focus on quantitative ureaplasma urine testing from genitourinary tract secretion samples.
- Versatility Redefined: Beyond clinical settings, our kit extends its applications to reproductive health, providing a comprehensive diagnostic solution for diverse healthcare scenarios.
- Empowering Healthcare: Facilitating early detection and management, our kit empowers healthcare professionals to proactively address ureaplasma infections, leading to improved patient outcomes and enhanced reproductive health.
| Specification | Details |
|---|---|
| Sample Type | Genitourinary tract secretions |
| Limit of Detection (LOD) | 6.62×10^2 copies/mL (8.80×10^2 CCU/mL) |
| Quantitative/Qualitative | Quantitative |
| Storage and Shelf Life | -20±5℃, 9 months |
How It Works:
Utilizing advanced Fluorescent Polymerase Chain Reaction (PCR) technology, the kit precisely amplifies and quantifies Ureaplasma urealyticum DNA. The fluorescent probes enable real-time monitoring of the PCR process, ensuring accurate and timely results. This innovative approach enhances diagnostic precision, allowing for early intervention and personalized patient care.
Key Techniques:
- Fluorescent PCR Technology: The kit employs state-of-the-art Fluorescent PCR technology, enhancing the sensitivity and specificity of Ureaplasma urealyticum DNA detection.
- Real-Time Monitoring: Fluorescent probes enable real-time monitoring of the PCR process, providing immediate and accurate insights into the presence and quantity of Ureaplasma urealyticum DNA.
- Quantitative Ureaplasma Urine Testing: The kit is specifically designed for quantitative ureaplasma urine testing from genitourinary tract secretion samples, allowing for precise and targeted diagnostics.
- Multiplexed Certification: Holding both CE and NMPA certifications, the kit ensures compliance with international and national quality standards, reaffirming its reliability and accuracy.
Embark on a new era of diagnostic precision with our Ureaplasma Urealyticum Fluorescent PCR Diagnostic Kit — where innovation meets certification for unparalleled diagnostic excellence.
Elevate Detection Precision with the Neisseria Gonorrhoeae Fluorescent PCR Diagnostic Kit:
Empower your diagnostic arsenal with the Neisseria Gonorrhoeae Fluorescent Polymerase Chain Reaction (PCR) Diagnostic Kit, a testament to innovation and precision in sexually transmitted infection (STI) diagnostics. With dual certifications from NMPA and CE, this kit redefines diagnostic excellence for Neisseria gonorrhoeae infections.
Distinctive Features:
- Dual Certification Assurance: Gain confidence in diagnostic outcomes with a kit that holds both NMPA and CE certifications, attesting to its compliance with international and national quality standards.
- Targeted Detection: Address the challenges posed by Neisseria gonorrhoeae infections with a focus on quantitative detection of Neisseria Gonorrhoeae nucleic acid from genitourinary tract secretion samples.
- Combatting STIs: As the second most common bacterial sexually transmitted infection (STI), gonorrhoeae necessitates a sophisticated diagnostic solution for prompt and accurate detection.
- Advancing Public Health: Contribute to public health initiatives by facilitating early detection and intervention, thereby reducing the morbidity associated with Neisseria gonorrhoeae infections.
| Specification | Details |
|---|---|
| Sample Type | Genitourinary tract secretions swab |
| Limit of Detection (LOD) | 500 copies/mL |
| Quantitative/Qualitative | Quantitative |
| Storage and Shelf Life | -20±5℃, 6 months |
How It Works:
Harnessing advanced Fluorescent Polymerase Chain Reaction (PCR) technology, this kit amplifies and quantifies Neisseria gonorrhoeae DNA with unparalleled precision. Real-time monitoring through fluorescent probes ensures immediate insights into the presence and quantity of the target nucleic acid, enabling timely clinical decisions.

Key Techniques:
- Fluorescent PCR Technology: The kit employs state-of-the-art Fluorescent PCR technology, enhancing sensitivity and specificity for the accurate detection of Neisseria gonorrhoeae DNA.
- Real-Time Monitoring: Fluorescent probes enable real-time monitoring of the PCR process, providing immediate and accurate insights into the presence and quantity of Neisseria gonorrhoeae DNA.
- Quantitative Detection: Tailored for quantitative detection, the kit focuses on Neisseria Gonorrhoeae nucleic acid from genitourinary tract secretion samples, ensuring precise and targeted diagnostics.
- Dual Certification: Holding both NMPA and CE certifications, the kit stands as a beacon of reliability, meeting the highest quality standards for diagnostic accuracy.
Revolutionize STI diagnostics with the Neisseria Gonorrhoeae Fluorescent PCR Diagnostic Kit — a convergence of certification and innovation for unparalleled diagnostic precision.
Revolutionizing Chlamydia Detection: Chlamydia Trachomatis Fluorescent PCR Diagnostic Kit
Elevate your diagnostic capabilities with the Chlamydia Trachomatis Fluorescent Polymerase Chain Reaction (PCR) Diagnostic Kit — a beacon of innovation certified by NMPA. Designed to address the challenges posed by Chlamydia trachomatis, the most common bacterial sexually transmitted infection (STI), this kit offers precise and qualitative detection from urethra or vaginal secretion samples.
Distinctive Features:
- Certification Credibility: Endorsed by NMPA, the kit adheres to stringent quality standards, ensuring reliability and accuracy in Chlamydia trachomatis detection.
- Targeted Qualitative Detection: Focus on qualitative detection of Chlamydia trachomatis from urethra or vaginal secretion samples, facilitating early intervention and reducing morbidity.
- Combatting STIs: Tackle the most prevalent bacterial STI with a diagnostic solution that emphasizes accuracy, efficiency, and timely detection.
- Public Health Impact: Contribute to public health initiatives by enabling comprehensive and reliable Chlamydia trachomatis testing, minimizing the impact of this pervasive infection.
| Specification | Details |
|---|---|
| Sample Type | Urethra or vaginal secretion |
| Limit of Detection (LOD) | 400 copies/mL |
| Precision | CV < 10% |
| Quantitative/Qualitative | Qualitative |
| Storage and Shelf Life | -20±5℃, 6 months |
How It Works:
Utilizing cutting-edge Fluorescent Polymerase Chain Reaction (PCR) technology, this kit amplifies and detects Chlamydia trachomatis DNA with exceptional sensitivity and specificity. Real-time monitoring through fluorescent probes ensures immediate insights into the presence of the target nucleic acid, facilitating timely clinical decisions.
Key Techniques:
- Fluorescent PCR Technology: Harness the power of advanced Fluorescent PCR technology for heightened sensitivity and specificity in Chlamydia trachomatis DNA detection.
- Real-Time Monitoring: Fluorescent probes enable real-time monitoring of the PCR process, providing immediate and accurate insights into the presence of Chlamydia trachomatis DNA.
- Qualitative Detection: Tailored for qualitative detection, the kit emphasizes the identification of Chlamydia trachomatis from urethra or vaginal secretion samples.
- NMPA Certification: Backed by NMPA certification, the kit stands as a symbol of adherence to rigorous quality standards for reliable Chlamydia trachomatis diagnostics.
Embrace precision and reliability in Chlamydia trachomatis testing with the Chlamydia Trachomatis Fluorescent PCR Diagnostic Kit — an innovative solution at the forefront of STI diagnostics.Fluorescent